WHAT'S NEW
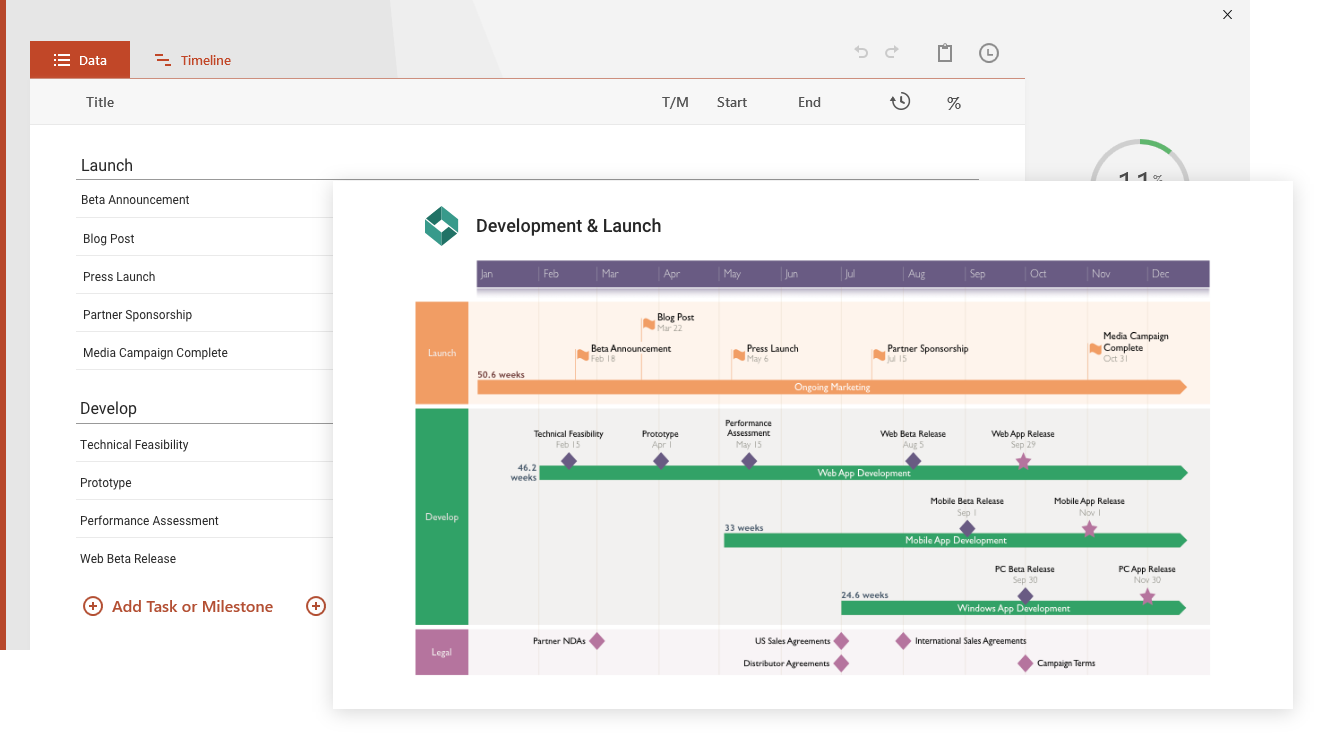
Office Flowchart
BetaDesign professional flowcharts in minutes, not hours, with our native PowerPoint add-in.
See more detailsOffice Plan
BetaGet instant insights and stay on top of project timelines and dependencies directly in Excel.
See more details